Estimated reading time: 12 minutes
More than eight million people in the U.S., almost 4% of the population, are estimated to suffer from gout. The disease often causes severe pain and physical disability.
Gout is associated with increased blood levels of uric acid, called hyperuricemia.
Uric acid is a biochemical substance the body has to deal with through our lifetime. It is an end product of the metabolism of compounds called purines.
Purines are abundant in nature. They are found in every living organism, from viruses to human beings. Purines are one of two families of nitrogen-containing molecules called nitrogenous bases; the other is called pyrimidines. Nitrogenous bases are used to construct the genetic material in every living organism, and they are an essential part of deoxyribonucleic acid (DNA).
Most mammalian species can use an enzyme called urate oxidase to convert uric acid to allantoin, a highly soluble product that is readily excreted in urine. Hence, these animals have very low blood levels of uric acid and therefore don’t develop gout.
By contrast, as humans do not possess uric oxidase, uric acid is the end product of purine metabolism. Thus, there is risk of uric acid accumulation with subsequent hyperuricemia and increased risk of gout.
The ionized form of uric acid, urate, is present in blood; approximately 98% exists as monosodium urate.
Uric Acid Production and Disposal
The liver is the primary site of uric acid production in humans. The production is the result of the breakdown of dietary purines and purine compounds produced by the body.
Uric acid generation involves the breakdown of purine mononucleotides into the purine bases, guanine, and hypoxanthine. These are then metabolized to xanthine. The final step is the oxidation of xanthine to produce uric acid. This step is catalyzed by an enzyme called xanthine oxidase.
Because uric acid is the final product of purine breakdown in humans, no further metabolism is possible. To avoid the accumulation of urate in the body, it has to be excreted. The primary pathway for excretion is through the gut and the kidneys.
Bacteria in the gut can degrade uric acid. This process which is called intestinal uricolysis is responsible for approximately one-third of urate disposal. Very little urate is found in stools, suggesting that uric acid is almost completely degraded by intestinal bacteria.
More than 70% of urate is excreted by the kidneys (1)
The kidneys’ handling of uric acid is a complicated process. Recent molecular cloning and genome-wide association studies have identified a host of urate transporters that provide critical new insight into the molecular mechanisms of urate transport. URAT1, GLUT9, ABCG2, NPT1 AND NPT4 are examples of urate transporters involved in the reabsorption and secretion of urate through the kidneys (2).
Urate excretion is enhanced in women of childbearing age due to the effects of estrogenic compounds. Hence, men usually have a much larger uric acid pool than women.
Uric Acid Reference Range
Serum urate concentrations in most children range from 3-4 mg/dL (178-238 µmol/L). During male puberty, levels begin to rise. Female levels remain low until menopause.
Adult men have mean serum urate levels of 6.8 mg/dL (404 µmol/L), and premenopausal women have mean serum urate values of 6 mg/dL (357 µmol/L). Values for women increase after menopause and approximate those of men. Throughout adulthood, concentrations rise steadily and can vary with height, blood pressure, body weight, renal function, and alcohol intake.
If mg/dL is the unit used like in the USA, the reference range for uric acid is
1.9 to 7.5 mg/dL for women
2.5 to 8 mg/dL for men.
If µmol/L is the unit used like in Australia, Canada and Europe, the reference range for uric acid is
113 – 446 µmol/l for women
149 – 476 µmol/l for men.
However, the values may vary based on the lab doing the testing.
Hyperuricemia
Hyperuricemia is a word use to describe elevated levels of uric acid in blood. Although there is no universally accepted definition of hyperuricemia, it is often considered present if uric acid levels are above 6.8 mg /dL (404 µmol/l) which is the approximate limit of urate solubility.
Decreased excretion of uric acid through the kidneys is responsible for about 85-90 percent of hyperuricemia. The remaining 10-15 percent are due to overproduction of uric acid.
Chronically elevated uric acid levels predispose some individuals to develop gouty arthritis, kidney stones, and uric acid renal disease. However, many people with hyperuricemia are asymptomatic.
Asymptomatic hyperuricemia is a stage in which the serum urate level is raised, but symptoms of gout are not present.
Gout
Gout is a term used to describe the clinical picture associated with the deposition of urate crystals within different body compartments. Hence, gout can be regarded as a disease of monosodium urate crystal deposition. It is reflected by high urate concentrations in blood, usually exceeding 6.8 mg /dL (404 µmol/l).
Gout has a substantial effect on physical function, productivity, quality of life, and health care costs. Uncontrolled gout is associated with significant use of emergency care services.
Women are less likely to have gout than men, but in the postmenopausal years, the gender difference in disease incidence decreases. Compared with whites, and/or ethnic minorities, especially blacks, have a higher prevalence of gout (3).
Although hyperuricemia is the underlying condition for the development of gout, many individuals with hyperuricemia never experience symptoms of urate crystal deposition. So, although hyperuricemia is a necessary predisposing factor for gout, the majority of hyperuricemic patients never develop gout (4,5).
The most common conditions associated with hyperuricemia are:
- recurrent attacks of acute gouty arthritis (joint inflammation), usually termed acute gout
- deposition of tophi in soft tissues (tophaceous gout)
- renal disease
- uric acid kidney stones
Apart from male gender and ethnicity, risk factors for gout include obesity, diets rich in meat and seafood content, alcohol-containing beverages, sodas and fruit juices high in fructose or sucrose content, hypertension, thiazide or loop diuretic use, and chronic kidney disease.
Acute Gouty Arthritis (Acute Gout)
The primary manifestation of gout is an exquisitely painful arthritis commonly termed acute gout. It usually affects a single joint (monoarthritis) but may sometimes involve many joints (polyarthritis) at the same time. Attacks usually last for a limited period but are typically replaced by intervals of freedom from all symptoms.
Acute gouty arthritis is predominantly a disease of the lower extremities. Involvement of the great toe (podagra) is very common. Other common sites are the insteps, ankles, heels, knees, wrists, fingers and elbows.
Commonly, the major attack begins at night, is extremely painful, and may sometimes be triggered by specific events such as trauma, alcohol ingestion, certain drugs or dietary excess.
Attacks of gouty arthritis may last only a day or two or up to several weeks but characteristically subside spontaneously.
Tophi
If the urate pool of the body continues to expand, monosodium urate deposits may occur in cartilage, tendons and soft tissues. These deposits, called tophi, are usually associated with chronic inflammation. A classic location of a tophus is the helix or antihelix of the ear.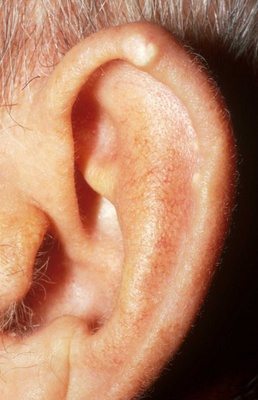
After treatment of hyperuricemia became more frequent, only a minority of patients develop visible tophi.
Gouty Renal Disease and Kidney Stones
Some renal dysfunction is common among patients with gouty arthritis. Several types of renal damage associated with hyperuricemia have been described. These are commonly described by the term urate nephropathy.
Increased urinary excretion of uric acid may lead to the formation of uric acid stones that may lodge within the urinary tract.
Other Associated Conditions
Obesity, high blood levels of triglycerides and hypertension are common among patients with gout. There is also increased prevalence of diabetes and atherosclerotic cardiovascular disease.
Treatment of Acute Gout
Although acute gout usually resolves completely within a few days, it is an immensely painful condition.
The primary goal of therapy is immediate relief of pain and disability.
When the acute attack has resolved the patients enters a symptom-free interval. However, flares of acute gout recur in the vast majority of patients. Hence, the secondary goal of therapy is to prevent recurrence.
NSAID’s
Several classes of anti-inflammatory agents are available for the treatment of acute gout. These are nonsteroidal anti-inflammatory drugs (NSAIDs), colchicine, systemic and intraarticular glucocorticoids, and biologic agents that inhibit the action of interleukin (IL)-1 beta (6).
Treatment is most effective if started early after the onset of symptoms. It should continue for the duration of the attack. Complete cessation of treatment can usually be done within 2-3 days after symptoms have resolved. However, slower tapering of therapy is needed if corticosteroids are used.
Administration of NSAID’s is the most commonly used treatment. It is usually effective if initiated within 48 hours from the onset of symptoms. Naproxen (500 mg twice daily), indomethacin (50 mg three times daily) and ibuprofen (600 mg three times daily) can all be used.
Typically, the duration of NSAID therapy for an acute attack is five to seven days. The drug can usually be discontinued one or two days after symptoms have resolved.
NSAID’s should be avoided in patients with reduced renal function, active duodenal or gastric ulcer, cardiovascular disease, NSAID allergy or ongoing treatment with anticoagulants (blood thinners).
Colchicine
Oral colchicine may be used if NSAID’s are contraindicated. As with NSAIDs, colchicine is most effective if administered early after the onset of symptoms.
The US Food and Drug Administration (FDA) recommends a schedule for the first 24 hours of colchicine treatment for acute gout with an initial dose of 1.2 mg of colchicine, followed one hour later by 0.6 mg, for a total dose on the first day of therapy of 1.8 mg. Another alternative is to administer 0.5-0.6 mg three times daily for the first day of treatment.
Colchicine is then continued for the duration of the attack, usually, at reduced doses, 0.6 mg once or twice daily as tolerated.
Colchicine must be administered carefully in patients with renal or liver impairment. Common side effects include diarrhea and abdominal cramping
Glucocorticoids
If NSAID’s or colchicine can not be used, glucocorticoids may be administered.
Direct intraarticular injection with glucocorticoids may be used. However, it should be avoided unless the diagnosis of gout is secured and infection can be excluded.
Glucocorticoids can also be administered orally. Usually, prednisone in doses of 30 to 50 mg once daily or in two divided doses is given until symptom start to resolve. This is followed by tapering of therapy, usually over 7 to 10 days.
Prevention of Recurrent Gout
The long-term goal of therapy of gout is to prevent recurrences of acute gout and reverse signs of disease, most effectively by reducing uric acid levels.
Although drug therapy is helpful in treating hyperuricemia, lifestyle modification is a critical issue in patients with gout. Treatment should target comorbid diseases such as hypertension, renal impairment, cardiovascular disease and different components of the metabolic syndrome (7).
Lifestyle Modification and Recurrent Gout
Purine restricted diets are traditionally recommended. One study found an association between purine-rich foods such as meat, seafood and purine-rich vegetables and gout in men (8).
However, it appears that purine restricted diets lead to very modest reductions in uric acid levels. On the other hand, a diet which emphasized caloric restriction, limited refined carbohydrates and saturated fats but allowed increased proportion of protein, led to substantial weight loss and a significant reduction in serum urate (9).
Frequent consumption of sugar-sweetened beverages or fructose-containing foods enhances the risk of having gout (10,11).
One study found that the intake of cherries reduced the risk of recurrent gout attacks compared with no cherries (12). However, these findings have not been confirmed in a randomized trial.
High intake of alcohol is associated with increased risk of recurrent gout.
One study found that episodic alcohol consumption regardless of type of alcoholic beverage (beer, liquor, and wine) was associated with an increased risk of recurrent gout attacks. It was concluded that individuals with gout should limit alcohol intake of all types. (13).
Several drugs may increase the risk of gout. For example, thiazide diuretics that are commonly used drugs for hypertension may elevate uric acid and lead to gout (14).
Treatment with aspirin has been associated with elevated levels of uric acid. Although this may have practical implications, particularly among elderly patients (15), discontinuation of aspirin is usually not required.
Drug Therapy for Recurrent Gout
Often, lifestyle modification and risk reduction strategies will not bet enough to prevent the recurrence of gout. Therefore, pharmacologic therapy that aims at reducing uric acid levels is often prescribed. Such drugs work by either by enhancing renal excretion of uric acid (uricosuric agents) or by decreasing urate synthesis (xanthine oxidase inhibitors) or both.
It has long been advocated that urate-lowering therapy should not be started until after an acute gout flare has resolved. Hence, waiting at least two weeks after the acute flare has subsided to initiate urate-lowering medication is recommended because acute urate-lowering can precipitate a gout attack and may worsen or prolong inflammatory arthritis (16).
Allopurinol, a xanthine oxidase inhibitor, is the most commonly used urate-lowering agent. A typical starting dose is 100 mg daily. Lower starting doses are recommended if renal insufficiency is present. A standard maintenance dose is 300 mg daily although the dose range may vary between 100-800 mg daily. Serum urate levels begin to fall within two days of allopurinol administration and reach stable levels in one to two weeks (17).
Febuxostat is another xanthine oxidase inhibitor that is available for treating recurrent gout.
Uricosuric drugs increase the urine excretion of urate. Probenecid, although infrequently used, is the most widely used drug in this class. Other uricosuric agents, such as benzbromarone, have also gained more use recently.
The angiotensin II receptor antagonist losartan which is often used in the treatment of hypertension has a modest uricosuric effect.
Uricase (urate oxidase) is the enzyme that catalyzes conversion of urate to allantoin. Pegloticase and Rasburicase are examples of uricases that are administered intravenously to lower uric acid concentrations. These drugs may be used when other urate-lowering therapy fails.
The Bottom Line
Gout is a common disorder accompanied by high blood levels of uric acid (hyperuricemia).
The primary manifestation of gout is an exquisitely painful arthritis commonly termed acute gout. Involvement of the great toe (podagra) is common.
The primary goal of therapy is immediate relief of pain and disability.
When the acute attack has resolved the patients enters a symptom-free interval. However, flares of acute gout recur in the vast majority of patients. Hence, the secondary goal of therapy is to prevent recurrence.
Gout is associated with central obesity, hypertension, high blood levels of triglycerides, and diabetes.
Lifestyle modification, emphasizing a healthy diet and alcohol restriction is helpful.
Drugs are used to treat the symptoms of acute gout and to prevent recurrence.



How about drinking water? My bloodwork last doctor visit came back with various readings that were off from norm. I suggested to my doc that maybe I was dehydrated based upon my reading of the various causes of diseases. Drank lots of water for two weeks, retested and all results came back normal.
Body can’t properly clear Uric acid if you don’t drink water. Most people are not drinking near enough. Stop with the drugs!
I agree that proper hydration is important. Dehydration may worsen kidney function and will not help lowering uric acid levels.
However, of course patients with acute gout need more than a couple of glasses of water to relieve symptoms.
I believe that drinking water is just a home remedy for the illness especially if it is not in the acute stage. Water intake will be every essential in diluting uric acid and still have the capability of excreting the acid.
Have you looked into FODMAP foods as the reason for gout? My MD friend Dr Gerry did that when he started to have problems with gout. He found it complelely resolved to gout if he stayed away from FODMAP foods. Information regarding FODMAPs can be found at the Monash University in Melbourne Australia where they have been measuring FODMAP’s in foods. They have published booklets to show the amounts of them in certain food. Incidently cherries are high in FODMAP’s. Funny thing is that taking out FODMAP’s was what he recommended for me with my ongoing IBS. It has worked well.
One aspect of gout which is too often overlooked in guidelines and in practice is that most gout flares are initiated during sleep. The sleep connection has been known at least since Dr. Thomas Sydenham wrote about it in 1683. A recent study by Dr. Hyon Choi et al confirms Dr. Sydenham’s observation. It is a very important clue to the pathogenesis of gout whether symptomatic or not.
Most gout flares are a direct result of sleep apnea, and overcoming the sleep apnea can cure the gout. Although Kelley’s Textbook of Rheumatology lists respiratory insufficiency as a cause of acidosis leading to hyperuricemia, the hypoxemia of sleep apnea actually has three effects which can lead to an overnight gout flare in short order. Effect #1 is cellular catabolism in which adenosine triphosphate degradation is accelerated, leading to nucleotide turnover which culminates irreversibly in the transient cellular generation of excess uric acid fed into the blood , faster than any food would cause. Effect #2 is transient hypercapnia and acidosis, so that the blood can hold less uric acid in solution. Effect #3 is a long term deterioration of the kidneys’ glomerular filtration rate so that removal of uric acid from the blood is slowed. Thus, with sleep apnea there is an abrupt increase in the influx of uric acid in the blood, slowed efflux, and abruptly reduced storage capacity — perfect storm conditions for monosodium urate precipitation. Furthermore, after awakening and normal breathing is restored, the first two effects dissipate so that a blood test taken during waking hours misses their peaks. And if monosodium urate has precipitated recently, then the measurement of serum uric acid is greatly undervalued.
Gout experts should feel embarrassed for having missed this connection for so long, especially since gout has been reported to have so many of the same comorbidities already known to be consequences of long-term untreated sleep apnea (eg., cardiovascular diseases, diabetes, kidney disease, hypertension.) One of the first steps for treating gout should be screening and diagnosis for sleep apnea, followed by treatment of the sleep apnea where indicated. I know from my own experience and the experiences of others that overcoming sleep apnea can prevent additional inflammatory gout flares immediately and completely. Effects #1 and #2 don’t occur, and Effect #3 may reverse over several months of effective treatment for sleep apnea. More importantly, gout is an early warning of sleep apnea, which when heeded can lead to the early treatment of sleep apnea, thereby greatly reducing the risk for the development of sleep apnea’s later developing life-threatening consequences. Using a gout flare as a sentinel event leading to the diagnosis and treatment of sleep apnea can save lives along with saving joints.
Very interesting Burton.
Thanks for sharing your thoughts on the issue.
Gout correlates with insulin resistance, hypertension, hyperlipidemia, and fractional excretion of sodium.
It’s possible there is a common renal defect shared by uric acid and sodium excretion, and that the latter contributes to hypertension.
Further, the large intestine is capable of excreting 30% of uric acid. Some evidence suggests some microbiome mixes are more efficient at excreting uric acid.
https://www.ncbi.nlm.nih.gov/pubmed/25763991