Estimated reading time: 12 minutes
By now, all of us should know that we are in the throes of an obesity epidemic. Worldwide, over 500 million people are believed to be affected (1).
Obesity in children and adolescents is on the rise and so is type 2 diabetes. And, according to the media and the experts, this will have horrible consequences that ultimately will drench our health system. Pretty serious isn’t it?
But, is carrying too much weight so terrible? Well, that’s what obesity is, isn’t it? The state of being grossly fat or overweight. It’s why many professionals use body mass index (BMI)(2) to define obesity. However, such an approach is very unfortunate and terribly misleading.
Obesity as a disease is very far from being simply a weight issue. Rather, it is a complex disorder of metabolism, abnormal energy regulation, and pathological behavior of fat tissue accompanied by inflammation, insulin resistance and lipid abnormalities, ultimately increasing the risk of, or directly causing, diabetes and cardiovascular disease.
According to the World health Organisation (WHO), overweight and obesity are defined as abnormal or excessive fat accumulation that may impair health (3). Accordingly, excessive fat accumulation should not be defined as overweight or obesity if it does not adversely affect health.
One of the main reasons obesity is regarded as a far-reaching public health issue is the association with adverse cardiovascular health. It is very well known that obesity is associated with increased risk of type 2 diabetes, a known risk factor for heart disease. However, the fact that the cardiovascular complications of diabetes seem to precede the onset of clinical diabetes, suggests that rather than cardiovascular disease being a consequence of diabetes, both conditions may have a common cause, i.e. they spring from a “common soil” (4). This is the “common soil” hypothesis. But what soil are we talking about here? Well, it’s clearly something that may cause both diabetes and heart disease.
Why does the accumulation of body fat increase the risk of cardiovascular disease? Why do some individuals with obesity have a normal metabolism (“metabolically healthy obese”) while the majority does not, and why do some people without obesity have a metabolic pattern that is mostly associated with obesity (“normal weight obesity”)? The missing link may be a condition called adiposopathy or sick fat.
According to the American Society of Bariatric Physicians, obesity is defined as a chronic, relapsing, multi-factorial, neurobehavioral disease, wherein an increase in body fat promotes adipose tissue dysfunction and abnormal fat mass physical forces, resulting in adverse metabolic, biomechanical, and psychosocial adverse consequences (5). Sounds pretty complicated, but interestingly, this definition does not mention body weight at all. In this case, adipose tissue dysfunction would be synonymous with adiposopathy.
So, let us take a closer look at the role of adipose tissue and the consequences of adiposopathy. This might move us closer to the secret behind the common soil hypothesis.
Historical Aspects
In 1947, Professor Jean Vague from the University of Marseille was the first to recognize that the distribution of body fat may better predict metabolic abnormalities than excess fatness in general (6).
Vague defined two different body shapes. Android obesity or apple shape refers to the accumulation of fat in the upper body area. Gynoid obesity or pear shape relates to the accumulation of fat on the hips and thighs. The latter is more common among women than men (7). In a paper published 1957, Vague wrote that android obesity “not only is associated with premature atherosclerosis and diabetes, but it is also the usual cause of diabetes in the adult in 80 to 90 percent of the cases (8).”
It appears that fat that lies directly under the skin (subcutaneous fat) may be relatively harmless while fat around the internal organs, often called visceral fat, is more likely to be associated with metabolic abnormalities. Research suggests that obese individuals with excess visceral obesity have a higher risk of diabetes, lipid disorders, and cardiovascular disease than those with less visceral fat accumulation (9).
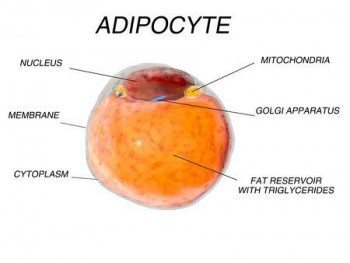
The discovery that the localization of adipose tissue might be of pathologic significance led to intensive research on the role of adipose tissue. It turned out that although the primary task of adipose cells may be to store fat, their role is much more extensive and complicated. For example, adipose tissue appears to be a major endocrine organ.
But, like every other organ in the body, adipose tissue may become diseased. Consequently, the term adiposopathy was defined and used to describe abnormal adipose tissue function that may be promoted and exacerbated by fat accumulation (adiposity) and sedentary lifestyle in genetically susceptible patients (10). Hence, the term sick fat.
From Healthy to Sick Adipocytes
The term adiposity is synonymous with obesity and refers to an excessive accumulation of fat in adipose tissue. Adiposopathy, however, refers to adipose tissue that is diseased and does not function properly, resulting in endocrine and immune responses that may cause metabolic abnormalities and directly promote cardiovascular disease.
Adipose cells are called adipocytes. Adipose tissue is made up of adipocytes, white blood cells, and fibroblasts, surrounded by connective tissue, collagen, nerves and blood vessels. Adipose cells constitute the majority of adipose tissue volume.
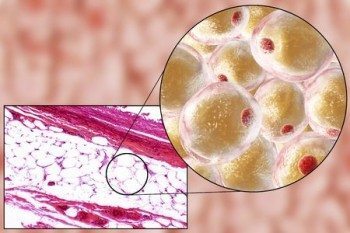
Previously, it was believed that we are born with a certain amount of adipocytes and that the production of new cells ceased early in life. However, although the number of fat cells appears to stay relatively constant throughout life (even after marked weight loss), indicating that the number of adipocytes is set during childhood and adolescence, the adipocyte number is continuously determined by the balance between the production of new cells and the removal of older cells. Research suggests that 10% of adipose cells are renewed in a year (11).
Adipocyte Hypertrophy, Inflammation and Insulin Resistance
When we overeat (positive caloric balance), adipose tissue may respond by using cellular signaling to call for the recruitment of more fat cells. By increasing the number of adipocytes (hyperplasia), adipose tissue may manage to store excess energy in the form of fat.
However, if the recruitment of new cells is insufficient, the remaining adipocytes will ultimately get larger, a phenomenon called hypertrophy. Hypertrophy may lead to adipocyte dysfunction that may provoke abnormal endocrine and immune responses and eventually lead to the metabolic abnormalities typical of sick fat.
One study showed that patients with type 2 diabetes had larger mean adipocyte size, more very large adipocytes, and lower total adipocyte number, independent of body weight (13).
In obesity, adipocyte hypertrophy and inflammatory responses are closely associated with the development of insulin resistance in adipose tissue. Recent data shows that lipid-overloaded hypertrophic adipocytes are insulin resistant independent of adipocyte inflammation (14). This may suggest that adipocyte hypertrophy may play a crucial role in causing insulin resistance in obesity.
The interplay between adipocyte hypertrophy and insulin resistance could be of crucial importance as insulin resistance is associated with substantially increased risk of developing cardiovascular disease, type 2 diabetes, high blood pressure, stroke, nonalcoholic fatty liver disease (15), polycystic ovary syndrome (16), and certain forms of cancer.
There is a lot of ongoing speculation as to why adipocytes become sick when they get larger. One theory is that large adipocytes may be more likely to suffer from lack of oxygen (hypoxia). In obesity, adipose tissue exhibits a major inflammatory response with increased production of inflammation-related proteins called adipokines. It has been proposed that hypoxia may underlie this inflammatory response and evidence that adipose tissue is hypoxic in obesity has been obtained in animal models (17).
The Role of Free Fatty Acids – Lipotoxicity Explained
Adipocytes store energy mostly in the form of triglycerides (18). Triglycerides are composed of three molecules of fatty acids attached to a glycerol molecule. If adipocytes are unable to store energy, free fatty acids may enter the blood stream (spill-over) which may have harmful effects on non-adipose tissue, affecting organs such as the liver, kidneys, pancreas, peripheral muscle, heart, and blood vessels. This phenomenon is termed lipotoxicity (19).
Lipotoxicity may play a role in the development of non-alcoholic fatty liver disease (20). It may lead to insulin resistance in peripheral muscle (21) and cause injury to pancreatic cells, possibly increasing the risk of diabetes (22).
It is believed that elevated systemic levels of fatty acids leading to lipotoxicity play an important role for many of the pathophysiological aspects associated with the metabolic syndrome.
Adipokines
Cytokines are regulatory peptides that mediate communication between cells. They are produced by a broad range of cells and appear to play a major role for the human body. Cytokines secreted by adipose tissue are called adipokines. Adipokines mediate cell signaling between adipose tissue and the immune, endocrine and cardiovascular system. Hence, adipose tissue may be regarded an endocrine organ as it secretes substances that control and regulate the activity of other cells and organs.
Adipokines play an important role in healthy energy metabolism and their effect on appetite and feeding behaviour influences the regulation of body weight. They also modulate immune functions and inflammatory processes.
Adipokines may affect other cells around them as well as remote organs such as the liver, heart, blood vessels, and peripheral muscle. Although they play a major role in normal bodily processes, they may also participate in various pathological situations, including obesity, type 2 diabetes, and cardiovascular disease.
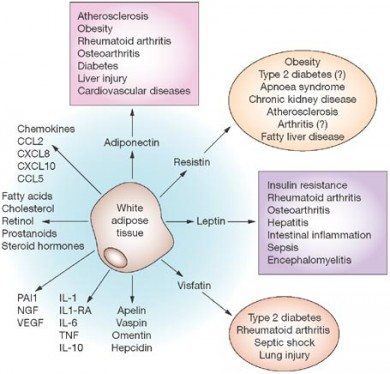
Currently, there are more than 600 known adipokines (23). Adiponectin (24), leptin (25), and resistin (26) are three of the best known.
Obesity is associated with low plasma concentration of adiponectin. Plasma levels of adiponectin are especially low in individuals with visceral obesity. It is believed that adiponectin deficiency may play an important role for many of the negative metabolic consequences of visceral fat accumulation.
Some adipokines may negatively affect health. For example, many adipokines are pro-inflammatory and may promote chronic low-grade inflammation (27,28). On the other hand, adiponectin is protective and appears to reduce inflammation.
Leptin is a 167 amino acid hormone synthesized by adipocytes. Leptin production correlates with of the amount of adipose tissue in the body. Obese people, therefore, produce more leptin than people with normal body weight.
Leptin receptors are found on a wide range of other cell types. Leptin regulates energy intake and feeding behavior through receptors within the brain (particularly in the hypothalamus) by providing feedback information about the status of energy reserves.
In other words, adipose tissue uses leptin to communicate with the hypothalamus. The signals can be twofold. Increased leptin production signals anorexigenesis; indicating the absence of hunger and that energy can be burned. Decreased leptin production signals orexigenesis; indicating hunger and that energy should be stored. It is the role of the hypothalamus to respond adequately to these signals.
Although leptin reduces appetite, obesity is associated with higher circulating concentration of leptin. Thus, obese individuals show resistance to leptin, similar to insulin resistance in type 2 diabetes. Hence, elevated leptin levels fail to control hunger and reduce appetite.
It is believed that normal adipokine production and secretion becomes affected when adipose tissue becomes sick. Sick fat shows signs of dysregulated production of adipokines. Increased production of IL-6, TNF-alfa and reduced production of adiponectin may promote inflammation and insulin resistance (28). Hence, abnormal or inappropriate secretion of adipokines may be an important link between sick fat and the metabolic abnormalities associated with obesity, type 2 diabetes, and cardiovascular disease.
The Take Home Message
Although body mass index is often used to define obesity, obesity as a disease is not primarily a weight issue. Rather, it is a complex disorder of behavioral patterns in combination with deranged metabolism, abnormal energy regulation, and unhealthy behavior of adipose tissue accompanied by inflammation, insulin resistance and lipid abnormalities, ultimately leading to increased risk of diabetes and cardiovascular disease.
The term adiposity is synonymous with obesity and refers to an excessive accumulation of fat in adipose tissue. However, adiposopathy (sick fat) refers to adipose tissue that is diseased and does not operate normally. Adioposopathy may be an important link between obesity and inflammation, insulin resistance, type 2 diabetes and cardiovascular disease.
One of the primary roles of fat cells (adipocytes) is to store fat during stages of positive energy balance. Fat tissue (adipose tissue) will initially respond by increasing the number of adipocytes (hyperplasia). However, if the recruitment of new adipocytes is insufficient, the remaining adipocytes will ultimately get larger, a phenomenon called hypertrophy. Adipose hypertrophy is characteristic of sick fat.
Adiposopathy is characterized by visceral adiposity, adipocyte hypertrophy, abnormal adipose tissue responses and increased release of free fatty acids which may cause lipotoxicity.
Adipose tissue produces a number of bioactive substances, known as adipokines, which during fat tissue expansion may trigger chronic low-grade inflammation and interact with a range of processes in many different organs. Although the precise mechanisms are still unclear, dysregulated production or secretion of these adipokines caused by excess fat mass and adipose tissue dysfunction can contribute to the development of obesity-related metabolic diseases.
It is very clear that obesity is associated with increased risk of type 2 diabetes, a known risk factor for cardiovascular disease. However, the cardiovascular complications of diabetes seem to precede the onset of clinical diabetes, suggesting that rather than cardiovascular disease being a consequence of diabetes, both conditions may have a common cause. This is the “common soil” hypothesis. That common soil may be adiposopathy.


Beautiful writing Dr. Sigurdsson. Any person with the so called “metabolic syndrome” is waiting for a breakthrough understanding of this complex interconnected scenario which is unfortunately currently treated only with “endpoints” such as cholesterol, glycemic driving multi (and sometimes conflicting) therapy.
Are we staring at the finger rather than looking at the moon?
Thanks for sharing.
Robert – Rome
Thanks Robert.
I agree. We tend to examine the finger rather than pointing at the moon.
Appreciate your input.
Excellent write up. I was fat, sick and inflamed. I lived with chronic pain for 10 years. Also had autoimmune issues. Fortunately not pre diabetic and my triglycerides were still okay. Changed my diet two and a half years ago and lost all the sick fat, stopped the autoimmune cascade and am a picture of health today…also 42kg lighter as a side effect…funnily enough today I eat double what I used to eat…obesity certainly is a complex issue. So glad I worked it out.
Hi Judy
Glad you managed to turn things around. Not an easy task I can tell you.
“…obesity is associated with increased risk of type 2 diabetes…”
As you know, statin use is also associated with increased risk of type 2 diabetes. In fact, this increase seems to be greater than any decrease in heart attack, etc.
A question Doctor. What do you think of restricted time eating diets?
A friend of mine went on one, lost 30 pounds in a few months, and kept on lifting weights and doing aerobic exercise pretty intensively. No noticeable adverse effects.
Thank you. David
David
Time restricted feeding as I see it is a type of intermittent fasting. I think it can be very useful if it’s done sensibly.
So what about Lipoedema – this is genetic, not caused by sudden increases in diet? Also calorie restriction doesn’t work to get rid of the fat deposits. Unusually obesity related risks seem less in women with Lipoedema? Lipoedema is in much need of investigation, I and millions of other women would love to hear your opinion about Lipoedema.