Recently, concerns have been raised about the increased risk of diabetes associated with statin therapy.
However, it has been pointed out that the risk of diabetes is low in absolute terms and when compared with the reduction in cardiovascular events achieved by treatment. In other words, the potential benefits to health are believed to outweigh potential risks. Therefore, expert guidance regarding the use of statins in clinical practice has not changed.
But is this how clinical medicine should be practiced?
From a public health perspective a therapy that increases the risk of one disease at the same time as it reduces the risk of another, may be acceptable, assuming the net effect will be positive in terms of general health risks.
However, from the individual perspective the picture may be different. We may not be willing to accept treatment that might slightly reduce our risk of heart disease but could increase the risk of diabetes?
What if we’re not one of the lucky ones. What if we get diabetes and don’t benefit in terms of cardiovascular risk. It’s a gambling game.
Furthermore, we still don’t know why statins increase the risk of diabetes. There appears to be a complex interplay between cholesterol metabolism, insulin resistance, obesity and diabetes that needs further clarification.
In fact, two recent papers may have cast some light on these issues.
So much is at stake. Millions of people are taking statin drugs; many of them will not derive much benefit, and some will be harmed. It’s time to let the cat out of the bag.
The Role of LDL Receptors
In 1985, Michael S. Brown and Joseph L. Goldstein were awarded the Nobel Prize in Physiology or Medicine for revolutionizing our knowledge about the regulation of cholesterol metabolism and the treatment of diseases resulting from elevated levels of blood cholesterol.
Brown and Goldstein found receptors on cell surfaces that mediate the uptake of low-density lipoprotein (LDL). LDL is the primary carrier of cholesterol in blood.
The liver plays a key role for the clearance of LDL from the blood stream. LDL receptors on the surface of liver cells bind LDL and remove it from the blood.
After binding to the LDL receptor, the LDL/LDL-receptor complex is moved to endosomes within the cells, where the LDL is released from the complex. The LDL receptor then moves back to the cell surface where it can bind to additional LDL particles. The free LDL left within the cells is transported to lysosomes and degraded into cholesterol ester droplets, free fatty acids, and amino acids.
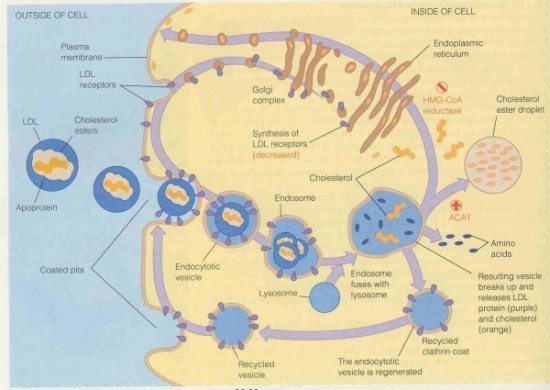
Statins and LDL Receptors
Statin drugs have revolutionized the pharmacological treatment of cardiovascular disease. These drugs effectively lower the levels of LDL cholesterol in blood.
Today, statins are widely used to treat patients with cardiovascular disease and healthy people with high blood levels of LDL cholesterol or other measures of increased risk.
Statins inhibit an enzyme called HMG-CoA reductase. This enzyme converts HMG-CoA into mevalonic acid, a cholesterol precursor.
Inhibition of HMG-CoA reductase by statins impairs cholesterol synthesis by liver cells and reduces circulating levels of LDL cholesterol.
Furthermore, the reduction of cholesterol within cells drives an increase in the expression of LDL receptors. This is believed to be a key step for the lowering of blood levels of LDL cholesterol achieved by statin treatment.
Familial Hypercholestrolemia and Diabetes
Familial hypercholesterolemia is a dominantly inherited disease characterized by high plasma levels of LDL cholesterol. Patients with this disorder are at increased risk for premature cardiovascular disease (1).
Brown and Goldstein discovered that the underlying mechanism to hereditary familial hypercholesterolemia is a partial or complete lack of functional LDL receptors leading to decreased clearance of cholesterol from the circulation and higher levels of blood cholesterol.
Interestingly, this is exactly the opposite to the situation with statins where cellular uptake of cholesterol is improved due to increased availability of LDL-receptors.
A new observational study from the Netherlands studied the risk of diabetes among 25,000 patients with familial hypercholesterolemia and 38,000 unaffected relatives participating in a national screening program between 1994 and 2014 (2).
The study found that the prevalence of type 2 diabetes was significantly lower in patients with familial hypercholesterolemia than their unaffected relatives.
The investigators believe that cellular cholesterol uptake is a common pathway in familial hypercholesterolemia and statin-induced diabetes. They propose that intracellular cholesterol accumulation associated with increased LDL receptor availability may be detrimental to the beta cells of the pancreas.
Their conclusion is supported by findings showing that addition of LDL cholesterol to isolated rat islet beta cells resulted in cell death (3). Another study found decreased glucose-stimulated insulin secretion in rodent pancreatic islets that were incubated with LDL-cholesterol (4).
Statins and Diabetes
A meta-analysis published 2010 that combined information from 13 individual studies (involving a total of 91,140 patients) showed that treating 255 patients with statins for four years led to one extra case of diabetes, whereas 5.4 cardiovascular events were prevented (5). Statin therapy was associated with a nine percent increased risk of diabetes.
Randomized clinical trials suggest that the risk of diabetes increases in a dose-dependent fashion (6).
Observational studies have also reported a higher risk of type 2 diabetes with statin treatment compared with individuals not taking statins (7).

It has been suggested that statins may affect insulin secretion and insulin sensitivity. One study has shown that statins may increase body weight which may be important for the risk of diabetes (8) .
A recent Finnish study of men with metabolic syndrome suggests that the risk of statin-induced diabetes may be higher than previously reported (9).
In this study, the investigators studied the effects of statin treatment on blood sugar and the risk for type 2 diabetes in 8,749 nondiabetic men age 45 to 73 years in a 6-year follow-up of the population-based Metabolic Syndrome in Men (METSIM) trial, based in Kuopio, Finland.
The mechanisms of statin-induced diabetes were addressed by evaluating changes in insulin resistance and insulin secretion.
Statin treatment was associated with an increased risk for type 2 diabetes even after adjustment for age, body mass index, waist circumference, physical activity, smoking, alcohol intake, family history of diabetes, and beta-blocker and diuretic treatment The hazard ratio (HR) was 1.46.
Thus, statin therapy appears to increase the risk for type 2 diabetes by 46% in men with metabolic syndrome.
Statin therapy was also associated with a significant increase in fasting blood sugar. Furthermore, individuals taking statins had a 24% decrease in insulin sensitivity and a 12% reduction in insulin secretion compared with those not receiving the drugs.
These new findings may have several implications.
Firstly, the results suggest that previous studies may have underestimated the risk of statin-induced diabetes.
Secondly, the findings demonstrate for the first time that increased insulin resistance is one of the mechanisms leading to diabetes in people receiving statins.
Thirdly, statins were found to affect insulin secretion from the pancreas. The authors suggest that impaired ability of the beta cells of the pancreas to respond to increased insulin resistance may be an important mechanism underlying statin-induced diabetes.
The Bottom Line
The increased risk diabetes associated with statin therapy is a concern although expert guidance regarding the use of these drugs in clinical practice hasn’t changed.
Two recently published papers have added important pieces to our understanding of statin-induced diabetes.
A study from the Netherlands shows that the risk of diabetes among patients with familial hypercholesterolemia is lower than in the general population
The study adds support to the theory that the expression and function of LDL-receptors may be important for glucose metabolism.
Improving LDL receptor function by statins may increase the risk of diabetes because intracellular accumulation of cholesterol may harm the beta cells of the pancreas, limiting their ability to produce insulin.
The ongoing trials on the new PCSK9 inhibitors will hopefully clarify some of the questions regarding LDL receptor function, glucose metabolism, and diabetes.
A recent Finnish study suggests that statin-induced diabetes may be more common than previously reported. This assumption is based on findings from a large observational study of men with metabolic syndrome.
It is likely that the prevalence of diabetes among statin users varies according to the population being studied. Individuals with metabolic syndrome usually have insulin resistance and are at increased risk of developing diabetes. Thus, their risk of statin-induced diabetes might be higher than in the general population.
It is estimated that around 70 million people in the US have metabolic syndrome or about one-third of all adults in the country (10). Many of these individuals are receiving statin treatment due to lipid abnormalities and increased risk for cardiovascular disease.
The study findings suggest that impaired ability of the beta cells of the pancreas to respond to increased insulin resistance may be an important mechanism underlying statin-induced diabetes.
According to a recent JAMA editorial, the recent evidence on the relationship between statin therapy and diabetes risk should not alter guidance regarding the use of statins in patients with elevated cardiovascular risk (11).
The evidence for the use of statins in secondary prevention is strong. Results of randomized clinical trials show that these drugs reduce the risk of future cardiovascular events and lower mortality in people with established cardiovascular disease.
However, the recently observed association between statin use and diabetes risk underscores the lack of knowledge about a a class of drugs prescribed to millions of people worldwide.
In my opinion, until we know more, a conservative approach is warranted when it comes to the use of statins in for healthy individuals without established cardiovascular disease.


Your article says: “…treating 255 patients with statins for 4 years led to 1 extra case of diabetes mellitus, whereas 5.4 cardiovascular events were prevented.”
I think they need to look closely at who those 1 in 255 patients that got diabetes were. What did they do/eat since on statins? Did they get adverse effects like muscle pain that influenced their ability to exercise and did they get lazy with their diets? Were they drinkers? Did they eat a lot of sugar? Vegetarian of big meat eaters? What were their cholesterol levels? How much did they drop after treatment? How did their HDL levels, LDL levels, triglycerides look before and after treatment with statins? Were they borderline diabetic or hypoglycemic prior to statin therapy? What statins did they take? What dose? My point: Maybe there is a way to eliminate increased risk of diabetes with statin therapy by looking at the patient before and after statin therapy.
Concerning the 5.4 cardiovascular events prevented, they need to look at those patient characteristics also. What were there various cholesterol components looking like before and after statin treatment. Were there any associations regarding weight or other factors on who benefited and who didn’t benefit?
Without this information, the statistics are meaningless to the patient, and the statistics can distort the factors in the decision to go with statin therapy or not. There seems to be enough usage of statins and a huge amount of money being spent on statins to motivate someone to spend some money to study these issues.
In my case, a male at age 66, I have no heart problems other than afib. I have done well on all stress/nuclear stress tests. My CAC score was 17. Blood pressure 110/70. My cholesterol hovers146 with HDLs of 53 plus. Based on 10 year CHD risk over 7.5% over 10 years (because of age and gender), my cardiologist put me on Pravastatin 10 mg 3 days a week . In two months my cholesterol dropped to 127 and HDLs stayed the same as did my blood sugar (80). My A1c was good. I exercise 6 days a week. I eat no red or fatty meat.
What did I accomplish with statin treatment?
Thanks for the article btw.
Pete
The 1 in 255 is not a specific patient. This is statistics, based on placebo controlled randomized trials. It means that if you treat 255 patients with statins you are likely to cause one case of diabetes which you would not get if you treated with placebo.
In individual cases, like yours, you can’t tell what statin treatment accomplished because there’s no way to know know how things would have progressed if you had not taken the drug.
@Pete
If your TC was 146 why did your doctor prescribe a statin 3x/week??
Have you come across any research on how statins may affect management of type 1 diabetes or other auto immune disease?
Axel,
Looking at the METSIM Finn study, I calculated the diabetes absolute risk increase to be 5.4% with a NNT of 1 in 18. Adjusting hazard ratios from 2.01 to say 1.46 (roughly 50% based on table 2 regression analysis) absolute risk now drops in half to 2.7% with a NNT of 1 in 37. If I did my math right…?
Either way the study suggests a huge associated diabetes risk using statins in primary prevention. Considering the NNT to prevent one heart attack (not death) using statins for primary prevention is say 1 in 150 it looks like patients are more likely to develop diabetes vs. preventing a non-fatal MI with statin use.
I agree that a conservative approach is best for primary prevention.
Got the same. 5.8% baseline risk, 46% is 2.7%, which is then the Absolute risk. Number Needed to Harm is 1/0.027= 37. So, you’d have to give 37 non-diabetics a statin for 5.9 years for 1 diagnosis of diabetes.
Hey, roboh98 thanks for verifying the math!
Jeff
I’ve looked at the numbers as well and agree with your assumption. The NNT for causing diabetes by statins in this group of patients appears low. It’s a real concern if it’s lower than the NNT for reducing cardiovascular events.
Jeff
The figures in the METSIM paper are quite interesting. Look at how late the curves separate. It appears that it takes a while before statin-induced diabetes develops. Obviously there is a risk that such an effect would be missed by a randomized clinical trial.
Great observation Axel. This longitudinal study provides some interesting data.
And by conservative approach do you mean no statin therapy?
Dr. Pearson, yes conservative approach for primary prevention generally means no statins here. Appropriate diet, CV imaging, advanced lipid testing, and CV inflammatory markers I find useful. The 2013 CV risk calculator I do not find useful.
Anthony
In my mind a conservative approach in primary prevention means only treating high risk individuals with statins, not all who have a ten year risk of CV above 7.5% according to a certain risk calculator.
However, there are certainly individuals with high risk, without known CVD where I would recommend statins.
Axel,
That seems a little vague. What, in your mind, makes an individual high risk (assuming no clinical coronary disease/stroke/PAD)?
Is it a higher 10 year risk? 15%?
Anthony
Yes, I know it seems vague. I’m not ready to rely on the risk calculators entirely. If I did, all almost all elderly individuals would end-up taking statins which I don’t think is right. I often use the risk calculator for support though and to explain the situation for my patients.
I rely more on clinical judgement and shared decision making. Many people prefer statins (over no statins) to reduce risk.
Here’s an example of a case where I would consider statins in primary prevention: A 55 year male with a hypertension, LDL 170 mg/dL (4.4 mmol/L) and family history of premature CAD (father had MI at age 57, two brothers had CABG in their sixties).
I could give you more examples but I think you know what I mean 🙂
Doc:
To answer your question, the reason that my doctor prescribed the statin is that, although my numbers look good, the fact that I am a 66 year old male, unless my HDLs were 55 or over, my 10 year CHD risk is over 7.5%.
The basis is as below:
“2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults”
https://content.onlinejacc.org/article.aspx?articleid=1879710
“A high level of evidence for an ASCVD risk-reduction benefit from initiation of moderate or high-intensity statin therapy in individuals 40 to 75 years of age with ≥7.5% estimated 10-year ASCVD risk was found .”
https://cvdrisk.nhlbi.nih.gov/evalData.asp
Age: 66
Gender: male
Total Cholesterol: 146 mg/dL
HDL Cholesterol: 53 mg/dL
Smoker: No
Systolic Blood Pressure: 110 mm/Hg
On medication for HBP: No
Risk Score* 8%
Means 8 of 100 people with this level of risk will have a heart attack in the next 10 years.
* Your risk score was calculated using an equation. Other NCEP products, such as printed ATP III materials, use a point system to determine a risk score that is close to the equation score.
I didn’t think HDLs of 53 was that bad. I don’t smoke or drink. No diabetes. BP 110/70. I have no prior history of heart disease.
So I don’t know what else I could do other than sex change or age reduction surgery, neither of which I would or could do. I think the guidelines are ridiculous. How many 66 year old males would NOT be prescribed statins using these guidelines? 5%? 95% can’t be having heart attacks in my age group.
What do you think? My doctor followed the guidelines.
Pete,
I have discussed this at length under my post “Should all men over age sixty take a statin drug?”(https://wp.me/p30zF0-cS). I advocate imaging for subclinical atherosclerosis by coronary calcium score or vascular CIMT/plaque and treating only those with significant atherosclerosis.
Dr. Anthony P.: Thanks for your comment. My CAC was 17 in 2014 and nothing showed up on my carotid artery scan. I don’t remember there being a score. When I asked why the statins then, the answer was “well you must have some plaque because your CAC wasn’t zero”. I even had something called a “gene expression test” that came out great. I’m positive the reason for the statin was the 8% 10 year risk. I exercise all the time and am very active. I don’t know anyone in as good shape at my age. I presume there are some older people who do marathons that might be super healthy using some criteria. However, I am not one of those emaciated runners. I’m medium build and well-muscled. Doctors always say” you are obviously in good physical shape”. So I don’t feel right about the statins. However, I am fastidious (maybe OCD is a better description) about what I eat and always have been, so I am susceptible to signing up for extra “insurance” by taking statins. I never know when I am over-thinking problems that don’t exist.
@Pete
Your Non HDL-C was 93 before statins. Your non HDL-C is now 73. If your doctor prescribed the statin based only on the 10 year CHD risk I might have a long talk with him
Charles, I hear you… I’m thinking maybe I need a different doctor. I heard that some of the doctors in the 13 person panel that developed the CHD 10 year risk calculator started to say they never meant for the calculator to be used to definitely prescribe statins… only that risk above 7.5% should start a discussion about diet, exercise, statins, etc. Personally, I think the calculator doesn’t work for my age group, but does for younger people. Thanks for your comment.
All very interesting… I would be very interested to hear any studies on how statins affect type 2 diabetics? My husband was put onto the One Treatment fFts all regime in NZ when he was diagnosed with Type 2 diabetes : Ace inhibitor, Metformin, Gliclazide, Statin and Aspirin (now off the Gliclazide and on Lantus) … His diabetes seems to be familial and he has Pacific Island heritage which also predisposes him to this… I have been worried all along as to the statins and wondered have they made his progression through the last 6 years onto insulin and worsening diabetes inevitable? … would he have progressed to this level and onto insulin anyway? Not sure but I think they need to do further studies on this? I heard that Pravastatin may improve insulin sensitivity whereas some of the other statins definitely make HbA1c levels higher. and may progress diabetes in people.. Do you have any interesting studies on diabetics and statins? Thanks 🙂 Love reading your articles… as an RN/GCN I have a strong interest in Nutrition and Epigenetics and Nutrigenomics… statins worry me a lot!
Jo
Oh and my CRP level was well below 1mg per liter as it always has been and I have no relatives with heart disease that I know of. My uncles lived into their 90s on both sides. My last living uncle just died at 97. All my blood tests were in normal range. I had tons of blood tests because my health plan is great and I don’t mind paying whatever I have to pay.
Dr. Gerber, you said “Appropriate diet, CV imaging, advanced lipid testing, and CV inflammatory markers I find useful. The 2013 CV risk calculator I do not find useful.”
So that means the only thing I can do, besides exercising moderately, is eat properly. The only thing my doctor SHOULD be doing is appropriate testing. If testing shows treatment is warranted, maybe statins would be appropriate.
@Peter
Bingo – the only thing I might suggest is a follow up CT Scan and carotid artery duplex to see if the plaque has progressed.
Your thoughts
“The evidence for the use of statins in secondary prevention is strong. Results of randomized clinical trials show that these drugs reduce the risk of future cardiovascular events and lower mortality in people with established cardiovascular disease.”
Being fairly new to the blog I think I probably missed where this had been covered earlier. Could you direct me to where it was talked about prior? I’m aware of the 4S study (and its limitations), is this where that statement came from?
And I think it important to note the difference between secondary and primary care – I think people don’t recognize the difference and think that a trial run for people with prior heart issues can be applied to people who haven’t had an issue. Ditto trial results when it comes to men and women.
Bob
Here are a few references that support the use of statins for the secondary prevention of cardiovascular disese.
https://www.ncbi.nlm.nih.gov/pubmed/7968073?access_num=7968073&link_type=MED&dopt=Abstract
https://www.ncbi.nlm.nih.gov/pubmed/8801446?access_num=8801446&link_type=MED&dopt=Abstract
https://www.ncbi.nlm.nih.gov/pubmed/9841303?access_num=9841303&link_type=MED&dopt=Abstract
https://jama.jamanetwork.com/article.aspx?articleid=201883
https://content.onlinejacc.org/article.aspx?articleid=1138915
https://www.ncbi.nlm.nih.gov/pubmed/12270864
https://www.ncbi.nlm.nih.gov/pubmed/19958964
https://www.nejm.org/doi/full/10.1056/NEJMoa061894
Just for the record, I am not for or against statins but rather about evidence being put into a proper context.
I think it is important to remember that type 2 diabetes (elevated glucose) for the vast majority of people is not a disease but a “risk factor” for cardiovascular disease (heart attack and strokes – which are the important clinical endpoints in contrast to glucose). When these studies say that statins cause type 2 diabetes what they really mean is that roughly 1-2% more people will have their glucose slightly pushed above an arbitrary threshold – the CVD risk associated with that small an increase in glucose is almost immeasurably small. In addition, if statins reduce CVD does it really matter if glucose goes up a bit. It’s exactly like the argument against HCTZ 20 years ago – HCTZ reduces CVD events and slightly increases glucose. Who cares, it reduces CVD which is why we worry about type 2 diabetes in the first place. Is there any evidence that statins cause “important” type 2 diabetes (big sugars and symptoms of hyperglycemia etc)? If there is then there may be an issue or am I missing something?
Given that only 1 of the 30 plus glucose lowering drugs on the market have any evidence that they reduce CVD (and even that evidence is weak) maybe we should stop measuring glucose all the time especially if they are on a statin because they are already on a drug that reduces CVD.
James
That’s an important and valid argument. I’ve had this discussion a number of times with my colleagues.
The first question is how do we define diabetes and of course that may vary quite a bit between studies.
In high risk individuals (secondary prevention and high risk primary prevention) we may accept that here’s a risk of affecting glucose metabolism. Whether we call it diabetes or not, a risk factor or a disease, is maybe not the main issue. It’s an unwanted adverse effect. On the other hand, considering the limited efficacy of statins in primary prevention, this problem can’t be ignored.
As you, I’m not for or against statins. However, I think they are important and useful drugs in many situations.
In general, I think there are two issues we have to strive to resolve. Firstly, many high risk patients who would benefit from statins are not receiving treatment. Secondly, many low risk individuals who will most likely not benefit are being treated with these drugs for years/decades.
Wow, this drives me crazy!
I’m on Atorvastatin for years now since I’m having heterozygous FH IIa.
I guess, for folks like me there’s hardly a chance to go along without statins but I wonder if I can at least lower the dosage a bit…but I’d like to get rid of ezetimibe first.
Thanks for this fascinating summary!!
Max
Max
Continuing with lipid lowering therapy may be very important in your case. So please don’t change your intake of these medications without consulting your physician.
Don’t worry, I won’t do it by myself. But I will at least question the therapeutic goals since my LDL is now well below 100 and I wonder if I’m not going too far down right now.
I have no cardiovascular troubles, there’s no history of CAD in my family and just a slight overweight is on my “must-change-List”.
But I’m afraid that my physicians are less informed and interested in this subject than me…
Max
@Max
What is your dosage of Atorvastatin?
Have you had a recent lipid panel done?
I’m taking 40 mg Atorvastatin and Ezetimibe daily.
My recent panel gave nice results but those are mainly caused by dietary intervention…I broke with all official recommendations!
Since November 2013 I’m well below 100 (= 2,6 mmol/L) and since I’m doing LCHF (lowCarb-highFat) I circle around 60 (= 1,56 mmol).
Without Ezetimibe and a less powerful statin (Simvastatin) I had 156-172 LDL cholesterol (= 4,06 – 4,7 mmol).
Before that, I used Cholestyramine only which caused a high variation of the LDL values (maybe a more direct dietary influence? I kept a poor diet then) – went up to more than 400 (10 mmol/L).
I’m just surprised to learn that high fat diet doesn’t influence LDL cholesterol (at least, under statins) but my Doc keeps warning that this doesn’t mean anything – the saturated fats may cause inflammation and all that despite good LDL values.
That’s why I want a check of C-reactive protein next time…
Max
It is untrue that the mechanism is unclear as to why statins have been linked to increase incidence of diabetes. Cholesterol is required for functioning of some of the glucose transporters (GLUT). Insufficient cholesterol —> malfunctioning or insufficiency of those glucose transporters, leading to sustained levels of elevated blood glucose.
Cholesterol and intermediates along the biochemical pathway inhibited by statins (HMG CoA-reductase inhibitors) are required to properly construct the “prenylated proteins” involved at least somewhat in insulin secretion from pancreatic beta-cells:
https://onlinelibrary.wiley.com/doi/10.1111/j.1582-4934.2007.00168.x/pdf
I think the sad part in all of this is that for many, dietary adjustments (contrary to those promoted by the diabetes foundations) can prevent T2d and in a number of instances reverse it. As with cholesterol, doing the opposite of what is generally advised seems to generate better results. (Your mileage may vary)
So the question with statins comes down to this: if eating fewer carbohydrates and a bit more fat can correct T2d and raise hdl while lowering tg and sometimes ldl why exactly are these drugs so profitable……and why accept the risks for a mediocre outcome long term?
@tw
What about the studies that show LFHC macrobiotic diets can reverse T2D in as little as 21 days?
tw
“why exactly are these drugs so profitable”
Because most people seem unable to follow a healthy diet. Be it low carb (there are, BTW, no long term studies of it in diabetes care), low fat, Mediterranean etc. etc.
It’s a tragedy, really, as the benefits of a healthy diet extend from T2D prevention to CVD, cognitive disorders, cancers etc. etc.
Axel, concerning the Finnish study (METSIM): as I cannot access the full-text version, could you comment on the doses used in the statin treatment? The risk was dose-dependent, and it’s been known for some time already that high doses of simvastatin (used by the study population) can be particularly problematic.
Mie
You should be able to download the paper here.
Here are the figures

Here are the figures
The full paper
https://www.schmidtlaw.com/wp-content/uploads/Cederberg-et-al-article.pdf
Increased risk of diabetes with statin treatment is associated with impaired insulin sensitivity and insulin secretion: a 6 year follow-up study of the METSIM cohort