The results from the recent GLAGOV trial suggest that aggressive lowering of LDL-cholesterol with a PCSK9-inhibitor on top of statin therapy in patients with coronary artery disease may reduce atherosclerotic plaque burden. Although these results provide support for the lipid hypothesis and may have significant implications for the future of cardiovascular medicine, our eyes are still on safety and long-term outcomes.
The lipid hypothesis implies that cholesterol, LDL-cholesterol in particular, is a key factor in causing atherosclerosis and coronary artery disease. In other words, it assumes that measures that elevate blood levels of LDL-cholesterol are bad and actions that lower it are good.
Epidemiological studies have shown that across cultures, cholesterol is linearly related to mortality from coronary artery disease (1).
However, when it comes to overall mortality, the issue is more complicated. Observational studies have shown a correlation between cholesterol levels and overall death rate in young and middle-aged people but not among the elderly (2,3). However, the mortality curve is J-shaped which means that those with the lowest cholesterol levels have increased mortality.
Furthermore, the relationship between cholesterol and mortality may be different for men and women. For example, a Norwegian study (4) found an inverse relationship between cholesterol levels and mortality among women. Hence, according to the authors, moderately raised cholesterol in women may not only be harmless but even beneficial.
The validity of the lipid hypothesis has also been tested in controlled interventional trials.
The Women’s Health Initiative (5) randomly assigned more than 48 thousand women, 50 – 79 years old, to a low-fat intervention or a comparison group. LDL-C was significantly lowered in the intervention group compared to the comparison group. Nonetheless, after six years of follow-up, there were no differences between the groups in the incidence of coronary artery disease and stroke.
The MRFIT trial (6) evaluated 12,866 high-risk middle-aged men who were randomly assigned either to a special intervention program consisting of treatment for high blood pressure, counseling for cigarette smoking, and dietary advice for lowering blood cholesterol levels or to their usual sources of health care in the community. Despite LDL-cholesterol being significantly reduced in the special intervention group compared to the “usual care” group, during a follow-up of seven years, there was no significant difference in total death rates between the groups and no differences in the number of deaths from heart disease.
The results of these two large trials indicate that lifestyle measures aimed at lowering LDL-cholesterol do not improve survival or reduce mortality from heart disease. However, drugs that lower the availability of atherogenic lipoproteins, either by decreasing their production or improving their clearance, might still provide clinical benefit.
Statins
Since their introduction more than 30 years ago, statin drugs have revolutionized the treatment of coronary heart disease. Randomized double-blind placebo-controlled trials have shown that statins reduce mortality and lower the risk of future cardiovascular events among people with established coronary artery disease.
However, their widespread use among healthy individuals for the purpose of prevention is still debated, and many experts have pointed out that side effects may be more common than previously believed.
Statins are potent inhibitors of cholesterol biosynthesis. Hence, according to the lipid hypothesis, most of the efficacy of statins is due to their cholesterol-lowering effects. However, the overall benefits observed with statins appear to be greater than what might be expected from changes in lipid levels alone, suggesting effects beyond cholesterol lowering.
The Jupiter trial (7) suggested that treatment with statins may have beneficial effects in people with relatively low LDL-cholesterol. The individuals who participated in this trial all had elevated levels of hs-CRP which is a marker of inflammation.
These results may suggest that the efficacy of statins may be explained by other mechanisms than cholesterol lowering, such as reducing inflammation. The cholesterol-independent or “pleiotropic” effects of statins include improving endothelial function, enhancing the stability of atherosclerotic plaques, decreasing oxidative stress and inflammation, and inhibiting blood clotting mechanisms.
PCSK-9 Inhibitors
The liver is the gatekeeper for low-density lipoprotein (LDL) and is responsible for its production and clearance.
Liver cells express specific receptors on their surface that bind LDL and remove it from the bloodstream. After binding to the LDL-receptor, the LDL/LDL-receptor complex is taken up by endosomes which are special compartments within the cells. The LDL receptor then moves back to the cell surface where it can bind to additional LDL-particles.
This process leads to removal of LDL-particles from the circulation and lower LDL-cholesterol levels. The free LDL left within the cells is transported to lysosomes and degraded into lipids, free fatty acids, and amino acids.
PCSK9 (Proprotein convertase subtilisin-like/kexin type 9) is a protein that regulates the expression of LDL-receptors in the liver.
PCSK9 is produced by liver cells and released into the blood stream. It binds to the LDL-receptor on the surface of liver cells, together with LDL. It also moves into the cell, together with the LDL-receptor/LDL complex.
After LDL is released from this complex, the LDL-receptor/PCSK9 complex is taken up by lysosomes for degradation, preventing the recycling of the LDL-receptor to the cell surface. Thus, PCSK9 is responsible for the degradation of LDL-receptors and therefore plays a critical role in the regulation of LDL-cholesterol levels.
Genetic mutations that lead to a loss of PCSK9 function are found in 1–3 percent of the population. These mutations are associated with very low LDL cholesterol levels and a lower incidence of CAD.
PCSK9-inhibitors are monoclonal antibodies directed against PCSK9. They lower LDL cholesterol by blocking the interaction of PCSK9 with the LDL-receptor on the surface of liver cells. This allows LDL-receptors to recycle to the cell surface, after releasing LDL within the cell, instead of being taken up and degraded in lysosomes. Increased concentration of LDL receptors on the surface of liver cells improves clearance of LDL, which will be reflected as lower levels of LDL-cholesterol.
Large-scale randomized clinical trials have shown that adding PCSK9 inhibitors on top of statin therapy in patients with established cardiovascular disease may provide some clinical benefit (8).
Can PCSK9-Inhibitors Reduce the Burden of Atherosclerosis?
Atherosclerosis is a key underlying component of cardiovascular disease. It is characterized by a chronic inflammation of the arterial wall, resulting from complex interactions between lipoproteins, white blood cells, the immune system, and several other factors.
Atherosclerotic lesions or plaques may cause arterial narrowing which may ultimately limit blood flow. Rupture of an atherosclerotic plaque may lead to thrombosis causing a sudden occlusion of the artery.
In theory, adding a PCSK9-inhibitor on top of statin therapy in patients with cardiovascular disease might reduce atherosclerotic plaques, reduce the risk of cardiovascular events such as heart attacks and strokes, and mortality may be reduced.
Studies have demonstrated good tolerability and efficacy of these drugs, with reductions in LDL-cholesterol ranging from 39-62 percent, compared with placebo, on top of maximally tolerated statin therapy and diet (9,10).
The big question is whether such a massive lowering of LDL cholesterol will provide clinical benefit and more importantly; will it cause harm? Unfortunately, these issues will not be answered until next year when the results from the large clinical trials become available (11).
However, recent evidence suggests that that the PCSK9-inhibitor evolocumab may indeed reduce atherosclerotic plaque volume. This is based on Amgen’s recent press release stating that the so-called GLAGOV study showed positive results (12).
GLAGOV
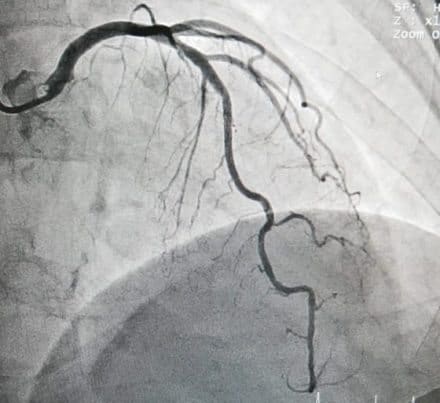
The GLAGOV study is a Phase 3, multicenter, double-blind, randomized, placebo-controlled trial designed to test whether treatment with the evolocumab in patients with established coronary artery disease modifies atherosclerotic plaque build-up in the coronary arteries of patients already treated with statins.
A total of 968 patients undergoing coronary angiography were randomized to receive either monthly subcutaneous injections of evolocumab 420 mg or corresponding placebo.
Intravascular ultrasound (IVUS) was used to quantify plaque volume in the coronary arteries. This is a high-resolution imaging tool that allows for the quantification of atherosclerotic plaques.
According to the press release, no new safety concerns were identified in the GLAGOV trial. The incidence of treatment-emergent adverse events was comparable among both groups. The primary endpoint was the change in percent atheroma volume from baseline to week 78 compared with placebo.
Currently, no further details are available on the results of the trial. These will be presented at the upcoming American Heart Association (AHA) Scientific Sessions in November 2016.
The GLAGOV results are concordant with the lipid hypothesis, suggesting that aggressive lowering of LDL-cholesterol will halt the atherosclerotic process. However, it has to be kept in mind that PCSK9-inhibitors affect several other lipid parameters. For example, treatment with evolocumab significantly reduces blood levels of lipoprotein (a) which may have significant clinical implications (13).
GLAGOV is the first randomized placebo-controlled trial showing a positive effect on atherosclerotic plaque volume by adding a PCSK9-inhibitor on top of statin therapy in patients with coronary artery disease. Whether this transforms into clinical benefit remains to be seen. Despite these promising results our eyes must still be on long-term efficacy and safety concerns.
So, until more data is available, GLAGOV may reflect new hopes for the future but could also turn out to be nothing more than empty promises.

Hi Axel. Wasn’t the difference in LDL-c between the arms in WHI just below 0,1 mmol/l after 3 years? This suggests poor adherence (and that the control group changed behavior as well), so the lack of effects of the intervention may not be surprising.
Besides, did you see this new paper in JAMA? Association Between Lowering LDL-C and Cardiovascular Risk Reduction Among Different Therapeutic Interventions. It suggests that all therapies that lower LDL-C by upregulation LDL receptors (including diet) reduces the risk of cardiovascular events.
Erik
You’re right about WHI. The difference in LDL-C was very small but it was statistically significant. Interestingly, by year 6, mean fat intake decreased by 8.2% of energy intake (SFA by 2.9%) in the intervention vs the comparison group which is quite a lot.
So, I assume we can agree that in this large trial, the low-fat dietary intervention failed. Of course the results might have been different with a more aggressive approach in order to achieve more reduction of LDL-C. But, that’s always going to be nothing more than a speculation.
Thanks for citing the JAMA paper. I hadn’t seen it. Very interesting. The non-statin intervention was a combination of diet, bile acid sequestrants, ileal bypass, and ezetimibe.
I don’t doubt the role of lipids in atherosclerosis but we have to acknowledge the inconsistencies of the lipid hypothesis and look out of the box because so much else matters.
For example, numerous studies of cholesterol lowering have failed to demonstrate a mortality benefit. The Mediterranean diet has consistently lowered cardiovascular events and mortality in many studies. However, this diet does not typically lower cholesterol levels.
The Results of the GLAGOV Study ere published in the JAMA today. Here’s the link to the paper https://jamanetwork.com/journals/jama/fullarticle/2584184
Getting rid of the cholesterol seems somewhat akin to throwing away the band-aids and then cutting yourself. Why not spend the time addressing why the cholesterol is having to do its job to begin with. The plaque is not just randomly placed, its there because of inflammation. NO???